In a 2007 season-opening
game, American football player Kevin Everett sustained a cervical C3-4
injury from tackling an opponent. While still in the ambulance, an
ice-cold saline solution was injected into Everett putting him into a
neuroprotective, hypothermic state and, later in the hospital, was
systemically cooled for several days through a cooling catheter inserted
in the femoral vein (see later discussion). In addition to this cooling,
Everett was treated 15 minutes post injury with the commonly used drug
methylprednisolone and had spinal decompressive surgery within seven
hours of injury. Although initially classified as having an ASIA-A
motor and se nsory
complete injury, he soon regained significant function, eventually
transitioning to an ASIA-D incomplete injury classification with the
ability to walk and normal bowel, bladder, and sexual functioning. Given
the various interventions, the degree to which his recovery can be
attributed to the cooling can not be determined.
nsory
complete injury, he soon regained significant function, eventually
transitioning to an ASIA-D incomplete injury classification with the
ability to walk and normal bowel, bladder, and sexual functioning. Given
the various interventions, the degree to which his recovery can be
attributed to the cooling can not be determined.
As reviewed in a number of articles listed in the
bibliography, studies dating back to the
1950s suggest that lowering central-nervous-system temperature can
mitigate the harmful effects caused by restricted blood flow (ischemia)
and associated oxygen deficiency (hypoxia) during operations that
disrupt blood flow to the brain or spinal cord. Based on these
observations and animal studies, cooling or hypothermic procedures were
developed to preserve neurological function after spinal-cord trauma.
Such procedures supposedly protect the injured
spinal cord by reducing the cord’s metabolic and energetic requirements
after injury. So to speak, it is like putting the injured neurons on
life support until they have a chance to recover. The cooled,
spinal-cord tissue doesn’t need as much viable cellular processes to
keep functioning and, as such, may survive longer after injury. Similar
to an ice-pack placed on a sprain, the cooling reduces neuron-damaging
swelling and bleeding at the injury site. The sometimes-used irrigating
cooling solutions may also wash out harmful toxins that accumulate after
injury and that promote secondary tissue damage.
Although studies cumulatively suggest that benefits
may accrue, care must be taken in over-generalizing results because the
studies 1) involved limited number of cases, 2) included no controls, 3)
reported only generalized improvement during a post-injury phase in
which some improvement is not unusual, 4) varied considerably in the
time lapsing from injury when spinal-cord cooling was started, 5) were
potentially confounded by the use of other drugs, and 6) weren’t always
limited to cases with traumatic SCI.
Most of the human clinical experience using
hypothermic procedures was acquired in the 1960-70s. By the 1980s,
enthusiasm “cooled off” because of ambiguous results, technical
complexity, and the decreased use of emergency laminectomy in acutely
injured patients, which was needed to expose the cord to cooling. [A
decompressive laminectomy is a surgical procedure in which various
function-compromising tissue or bone fragments that compress the cord
are removed]. Recently, with the development of more sophisticated
technology, the protective potential of post-injury, spinal-cord cooling
is being revisited.
Essentially, procedures can be categorized as
either systemic (i.e., whole body) hypothermia or localized cooling:
LOCALIZED COOLING
Reported in 1970, Dr. GastonAcosta-Rua (USA)
treated two men (17 and 21) with thoracic injuries from motor-vehicle
accidents with spinal-cord cooling.
After a decompressive laminectomy, the spinal-cord’s outer dura membrane
was opened, and the cord cooled for three hours with a re-circulated
saline solution (2o C). The time lapsing from injury to
cooling was two days in the first case and several hours in the second.
Although both patients improved, the author only stated that the
procedure “may contribute to the recovery of spinal cord function.”
In a 1971 article, Dr. Y. Demian et al described
treating three individuals (age 15, 17, and 18) with acute cervical
injuries. After a laminectomy surrounding the injury site, the
spinal cord was cooled for 1.5 – 3 hours with an ice-cold,
physiologically compatible, saline solution. In two cases, the time from
injury to cooling was about five hours; in one case, it was over 12
hours. Recovery was noted in all.
Summarized in a 1971 article, Dr. Robert Selker
(USA) used hypothermic cooling to treat four acutely injured
patients within three hours of injury (Surg
Forum 1971; 22). Two
had cervical injuries and two thoracic; three injuries were from
gunshot. Two patients died several months after the surgery, and the
other two regained some function.
In 1972, Dr. Dexter Koons et al (USA) reported
treating five patients with acute cervical (2) and thoracic (3) injuries
with hypothermic procedures.
Patient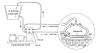 s
underwent a decompressive laminectomy 3-7 hours after injury. After the
spinal-cord’s outer dura membrane was opened, the cord was cooled with a
physiologically compatible saline slush for 30 minutes and the dura
closed. The majority of patients did not regain function.
s
underwent a decompressive laminectomy 3-7 hours after injury. After the
spinal-cord’s outer dura membrane was opened, the cord was cooled with a
physiologically compatible saline slush for 30 minutes and the dura
closed. The majority of patients did not regain function.
Reported in 1973, Drs. William Meacham and
Warren McPherson
(USA) treated 14 patients with spinal-cord cooling within
eight hours of injury. Age ranged from 16 to 56; all but three were male;
and 12 and 2 had cervical and thoracic injuries, respectively. Most
patients were also treated with neuroprotective steroids. A
decompressive laminectomy was carried out over three vertebral sections
surrounding the injury site, and the cord cooled by cold saline (4o
C) for three hours. Four patients died, although the investigators
believed that the deaths were not surgery associated. Of the 10
surviving patients, seven demonstrated some improvement, including
improved sensation, motor control, and bladder functioning.
In a 1975 article, Dr. Juan Negrin (USA)
described the treatment of three patients in the early to mid 1960s with
delayed hypothermic cooling.
After a cord-exposing, decompressive laminectomy, cooling was carried
out after opening the cord’s covering membranes (called subarachnoid
cooling) or leaving them intact. [As discussed in the appendix, the cord
is covered by three membranes: the outer dura mater, the middle arachn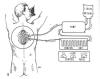 oid
membrane, and the innermost pia mater.] Alternatively, procedures were
developed to cool the spinal cord nonsurgically by routing the cooling
solution in and out of the cord through catheters.
oid
membrane, and the innermost pia mater.] Alternatively, procedures were
developed to cool the spinal cord nonsurgically by routing the cooling
solution in and out of the cord through catheters.
With the first patient, who sustained a thoracic
injury five hours before laminectomy, the spinal cord was cooled without
opening the membrane for three, 45-minute periods two, three, and four
days after surgery. No improvement was reported. With the second
patient, who had a laminectomy a day after sustaining a SCI from a fall;
the cord was cooled for one hour by subarachnoid cooling. Several weeks
later when the spinal cord needed to be cut open again, cooling was
carried out for a second time for another hour. Over time, the patient
regained considerable function. Due to delayed complications, the third
patient’s decompressive laminectomy was undertaken a year after
acquiring a cervical injury by a car accident. At that time,
subarachnoid cooling was carried out for 45 minutes. Improvement was
noted.
In a 1979 article, Dr. Charles Tator (Canada)
summarized his experience irrigating the acutely injured spinal cord of
11 patients treated over the 1968-77 period with either cooled or
body-temperature solutions. He
suggested that non-cooling irrigation may still provide benefits because
the physiologically supportive irrigating solution would provide oxygen
to the injured tissue, create a more biochemically supportive
environment, and flush out injury-created noxious substances. Seven and
four patients had sustained clinically complete cervical and thoracic
injuries, respectively; and age ranged from 16 to 56. The time lapsing
from injury to surgery varied from 3-8 hours. The irrigations were
carried out with the spinal cord’s dura (outer) and arachnoid (middle)
membranes widely opened. Six and five patients were irrigated with
hypothermic (5o C) and body-temperature (36o C)
solutions. Three patients recovered some sensation, one of whom
recovered some motor function (toe wiggling). Of the three, two and one
were irrigated with cooling and body-temperature solutions,
respectively.
In 1976, Dr. Albino Bricola et al (Italy) described the
hypothermic treatment of seven men and one woman with acute SCI (10).
Age ranged from 18 to 61 (average 33) years; four patients each had
cervical and thoracic injuries. The time from injury to initiating
cooling ranged from seven to 26 hours. After a wide, three-level
laminectomy, the cord’s covering dura-mater membrane was opened, and
then the cord irrigated for 10-20 minutes - the time needed to inspect
it for damage. Thereafter, the membrane was closed and catheter tubing
put over the injury area, through which a cooling solution flowed for a
duration ranging from 1.5 hours to eight days. Three patients died; four
of the five survivors recovered some motor and sensory function.
In 1984, Dr. Robert Hansebout and colleagues reported the results of treating seven male and three female
patients (6 thoracic and 4 cervical) within 8.5 hours of injury with
both spinal-cord cooling and neuroprotective steroids. After
decompression, a cooling “saddle” (6o C) was placed lightly
against the cord’s outer dura membrane for four hours. Followed for at
least six months, three of the patients accrued some motor or sensory
recovery (one died).

SYSTEMIC COOLING
Due to a rise in body temperature after injury,
Everett was systemically cooled, a procedure that has been more
extensively used in traumatic brain injury (TBI). Even with TBI,
however, the benefits of such cooling have been ambiguous. For example,
a study completed in 1998 involving 392 patients with TBI did not
demonstrate significant benefits, except in younger, quickly treated
patients.
For SCI, medications used to prevent shivering
interfere with monitoring neurological function and promote health
complications. Addressing this issue, Dr. Jogi Inamasu and colleagues
(Japan) stated “… patients with cervical SCI, who are most vulnerable to
respiratory infection, hypotension, and bradycardia [slow heart rate]
may be further compromised by induction of systemic hypothermia,”
further noting that the prolonged use of sedatives and muscle relaxants
essential during systemic hypothermia may worsen the respiratory
function of these “fragile patients.”
Nevertheless, because animal studies suggest that
post-injury elevated body temperatures are detrimental, Miami-Project
investigators have started using state-of-the-art technology to treat
acutely injured patients with mild hypothermia, producing a
several degree drop in body temperature.
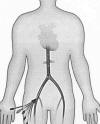
Basically, a catheter is placed in the patient’s blood vessel, and a
thermo-regulating device closely monitors and adjusts blood temperature
as it passes by the catheter. The study will follow long-term benefits
by assessing improvements in motor and sensory function and acquisition
of daily-living skills.
The cooling procedures were more thoroughly
described in a 2009 article, which summarized the hypothermic treatment
of 14 patients with complete cervical injuries. Patient age ranged from
16-62 years (average 39.4), and 10 were men and four females. The time
elapsing from injury to initiation of treatment averaged 9.2 hours.
After the cooling catheter was inserted into the femoral vein (see
illustration), patients were cooled at a maximum rate 0.5o C/hour
until the target body temperature of 33o C was reached, which
usually took less than three hours.
 The
target temperature was maintained for 48 hours, after which patients
were warmed at a rate of 0.1o C per hour. The duration of
total cooling was nearly four days.
The
target temperature was maintained for 48 hours, after which patients
were warmed at a rate of 0.1o C per hour. The duration of
total cooling was nearly four days.
Patients were sedated and given medications to
prevent shivering, although shivering with these cervical injuries was
already greatly attenuated due to virtually complete body paralysis. As
expected, lowering the body temperatures slowed the heart rate.
Respiratory complications were common and included atelectasis (collapse
of lung tissue) (12 patients), pneumonia (8 patients), and acute
respiratory distress syndrome (2 patients). The investigators noted,
however, that a control group of patients who did not receive
hypothermic treatment had comparable rate of respiratory complications.
No functional outcomes were reported by the investigators in this
article.
Clinical outcomes were reported in 2010 after the
subjects had been followed for approximately a year (ref). Although all
14 subjects initially had ASIA-A complete cervical injuries, the
injuries of six (43%) became incomplete after a year. Three improved to
ASIA B with some sensory improvement, two to ASIA C with partial
recovery of both sensory and motor function, and one improved to ASIA D
with even greater improvement. These hypothermia-treated patients were
compared to 14 untreated patients, who were matched with respect to
injury level and completeness, and age. Although improvement in the
cooled patients appeared greater than that observed in controls and that
reported in the literature for the natural history of SCI neurological
recovery, definitive conclusions could not be made because the study’s
small sample size could not generate statistically significant results.
The investigators concluded that the results, albeit lacking the needed
statistical power, were encouraging enough to warrant the initiation of
larger, more definitive studies.
In 2013, the investigators reported the results of
cumulatively treating 35 patients with their cooling procedures. As
before, all patients had Grade-A complete cervical injuries at the time
of admission, although four improved within 24 hours to the Grade-B
incomplete level. Of the 35 patients, 27 were males, and the majority
became injured from motor vehicle accidents. Excluding several patients
with delayed admission, the average time from injury to hypothermia
treatment was 5.8 hours. All but four patients were followed for 12
months. Of the 35 patients, 15 improved at least one grade after the
final follow-up assessment (only 11 if you exclude the four individuals
who had improved a grade within the first day). The overall results
compared favorably to what would be expected based on historical data.
Once again, a high incidence of complications, especially respiratory,
was observed, however, again noting that such complications are not
uncommon with cervical injuries.

POTENTIAL MECHANISMS OF
HYOPOTHERMIC COOLING
A recent article by Dr. W. Dalton Dietrich
and colleagues (part of the aforementioned overall Miami Project group
evaluating hypothermia) discussed several physiological mechanisms by
which hypothermia may protect CNS tissue after injury (15):
1) Metabolic Influences: Hypothermia lowers
cellular metabolic and energy requirements. It specifically lessens the
depletion of ATP, an extremely important molecule that is consumed to
drive most biochemical processes.
2) Blood Circulation: Hypothermic-induced
changes in blood flow may be neuroprotective.
3) Excitotoxicity: Cooling reduces
excitotoxicity. After injury, nerve cells lyse releasing excitatory
amino acids, such as glutamate, which soon reach toxic concentrations.
Through interactions with receptors on neighboring cells, excessive
glutamate will initiate a neurotoxic biochemical cascade.
4) Blood-Brain Barrier: The BBB restricts
the passage of various substances between the bloodstream and the CNS.
Injury compromises this barrier allowing for the passage of water and
neuron-damaging substances into the CNS. Hypothermia reduces this
injury-associated permeability.
5) Calcium: Calcium is an ion (i.e.,
electrically charged molecule) which is routinely present in extremely
low concentrations within nerve cells. Injury disrupts the calcium
equilibrium necessary for neuronal functioning; hypothermia lessens this
disruption.
6) Inflammation and Edema: Due to the
preservation of the blood-brain barrier, hypothermia lessens 1) the
infusion of inflammatory cells into the injury site and 2) edema
swelling cause by the fluid accumulation.
7) Neuronal Cell Death: Hypothermia
decreases post-injury apoptosis, a form of programmed
cell death common after injury.
8) Global Molecular Changes: Hypothermia
affects post-injury expression of genes and the substances they produce.
These alterations may be neuroprotective.
TOP